Select your region
-
Europe
-
Americas
-
Africa and Middle East
-
Asia Pacific
ABSTRACT
Background: Diaper dermatitis (DD) causes discomfort and emotional distress and creates possible sources of infection among newborn intensive care unit infants. Diaper dermatitis remains prevalent despite studies documenting an understanding of prevention and treatment modalities. Standardizing perineal skin care guidelines could reduce DD. Purpose: Implement perineal skin care guidelines, while introducing novel diaper wipes, to decrease the incidence of DD by 20% within a 1-year period.
Methods: Our unit reviewed evidence-based literature to develop a standardized perineal care guideline for prevention and treatment, encompassing the use of novel, preservative-free diaper wipes with grapefruit seed extract. The outcome measures were the incidence and duration of DD. Process compliances were monitored. The balancing measure was the rate of fungal skin infection while using preservative-free wipes.
Findings: Between July 2017 and March 2019, 1070 infants were admitted for 1 or more days, with 11% of those being born at less than 30 weeks of gestational age. After guideline implementation in January 2018, the incidence of DD decreased by 16.7%. The incidence of severe cases dropped by 34.9%, with 3.5 days per 100 patient-days shortened duration. Process compliance was achieved. Neonates tolerated the novel wipes without increased fungal skin infection.
Implications for Practice: The Perineal Skin Care Guidelines could reduce the rate and duration of DD. Newborn intensive care unit infants tolerated the novel diaper wipes.
Implications for Research: Additional research on wipes containing other types of extracts or ingredients.
Key Words: diaper dermatitis, diaper rash, grapefruit seed extract, infants, neonates, perineal skin care, premature, preservative-free chemical-free water wipes, preterm
Diaper dermatitis (DD), also referred to as diaper rash, describes the inflamed skin and lesions of the diaper area involving the lower abdomen, buttocks, and perianal and perineal areas. The reported incidence of DD varies from 21% to 25% among newborn intensive care unit (NICU) infants and up to 100% in the first 2 years of life.1-3 Signs of DD include erythema, excoriations, denuded skin, and bleeding. The resulting diaper area skin breakdown becomes a potential entrance for skin and systemic infection.4 In addition to infectious risk, infants with DD often display emotional and physical distress, which is evidenced by a higher salivary stress cortisol level.5 Parents are often distressed by DD, which may affect their perceived quality of the NICU care their infants received.6
The causes of DD are multifactorial. Infants, especially preterm infants, have an underdeveloped stratum corneum as the outer protective skin layer. Moisture from urine and digestive enzymes from alkalinized stool further irritate the vulnerable perineal skin. Attempts to remove these irritants with excess rubbing and use of preservative-containing wipes may disrupt skin barriers.7 NICU infants who might be exposed to antibiotics, fortified diets, and prenatal illicit drugs were at a greater risk for DD due to altered gut flora, stool composition, and stooling frequency.4,8
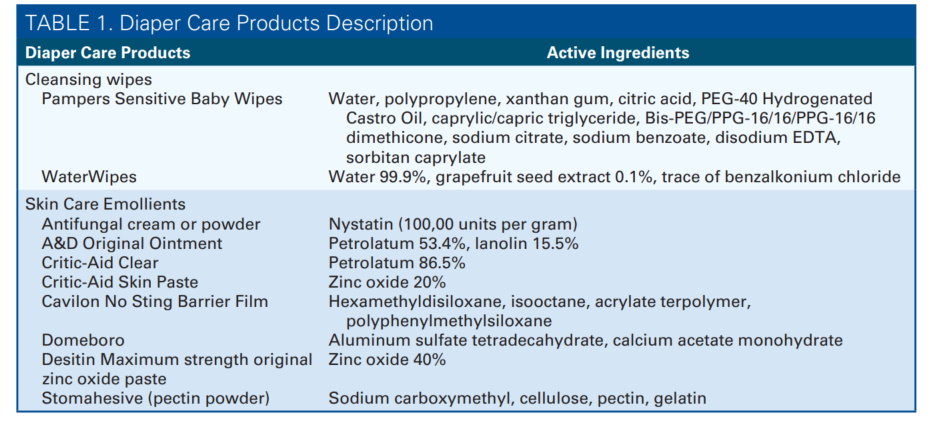
A substantial body of literature has reviewed the prevention and treatment of DD.1-11The advocated management included removing skin irritants, gentle cleansing, using skin emollients, and appropriate diapering. Despite a decent understanding of the management, DD remains prevalent in the NICU.2 The issue may be due to the fact that most NICUs lack a standardized approach to perineal skin care.1 For example, skin assessment and reporting of DD are usually inconsistent, which leads to underestimating the prevalence rate and poses challenges in monitoring care improvement.2 In addition, some of the commercially available diaper wipes contain additives that can provoke sensitive skin without the caretaker’s knowledge (Table 1).12 Cleaning with cotton wool or cloth with water seems to be more untainted, but multiple comparative studies have shown that diaper wipes are more beneficial.13-15 The pH buffers in diaper wipes may counteract alkaline urine and restore an appropriate skin pH balance.13 Also, the wipes contained surfactants as a gentle cleanser, which helps remove the fatty portion of feces.4 Thus, it is crucial to choose the most appropriate wipes. Besides wipes, choosing from numerous barrier emollients can be overwhelming. Different barrier emollients have various ingredients including petroleum and zinc oxide (Table 1), as Malik et al2 reported that up to 5 different skin care products have been used within the same unit. These nonstandardized prevention and treatment approaches can be confusing to caretakers. Studies have shown that the implementation of evidence-based practice guidelines was successful in reducing DD.1
The key element to reduce DD is establishing a prevention-focused guideline. Our NICU, at the University of Utah Hospital, experienced a high incidence of DD. Thus, a multidisciplinary quality improvement team was formed and tasked with identifying key drivers, reviewing current evidence-based literature, redesigning skin care guidelines, implementing changes, educating staff, tracking progress, and monitoring compliance. Our Perineal Skin Care Guidelines standardized skin assessment and documentation, introduced novel wipes, and focused on prevention and treatment with emollients. The aim was to decrease the incidence of DD by 20% within a 1-year period.
METHODS
Setting
The University of Utah’s NICU was a level III NICU with an average census of 35 patients per day. Infants who required oxygen less than 1 L, received less than a dextrose 12.5% fluid infusion for hypoglycemia, or were more than 34 weeks of birth gestational age (GA) would be transferred out of the NICU to a step-down unit. Infants requiring surgery were also transferred out to an affiliated surgical NICU.
Study Population
The project targeted infants who were admitted to the NICU between July 2017 and March 2019. The infants admitted from July 2017 to December 2017, before guideline implementation, served as the baseline for the incidence and duration of DD. The infants’ GA, birth weight (BW), birth month, skin assessment documentation, incidence and duration of DD, length of stay (LOS), and skin emollients used were collected. The same information was collected during guideline development and postimplementation. The inclusion criteria were all infants admitted and stayed for more than 1 day in the NICU. The exclusion criteria were infants who were transferred out of the NICU or died within the first day of life.
Intervention
A multidisciplinary quality improvement team was formed to address infants’ perineal skin integrity issues. The team consisted of bedsides nurses, charge nurses, occupational therapists, and attending physicians.
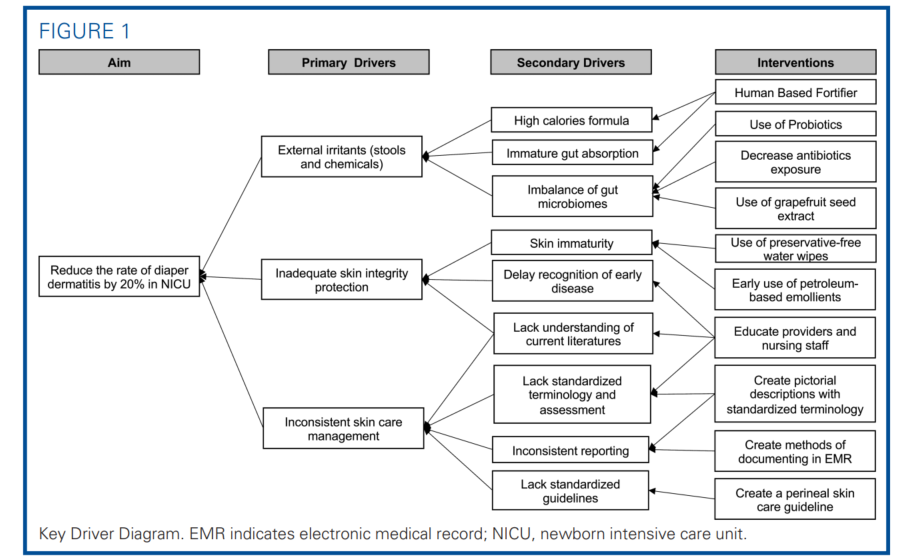
Using the Key Driver Diagram tool (Figure 1), the team identified the neonatal risk factors of DD and compiled potential interventions aimed at reducing DD based on literature review, experts’ opinions, and multidisciplinary consensus. One of the risk factors was external irritants including chemical irritants and stools. Our NICU had been limiting formula exposure and using more donor human milk and human milk–based fortifiers. We also attempted to restore healthy gut microbiomes with giving probiotics and decreasing antibiotics exposure, though DD was still prevalent. Inconsistent skin care management and inadequate use of diaper skin integrity protection were also identified as primary drivers. Thus, the team developed the Perineal Skin Care Guidelines to standardize skin assessment, documentation, prevention, and treatment (Figure 2). The published AWHONN’s Neonatal Skin Care Evidence-Based Clinical Practice Guidelines and Heimall’s Perineal Skin Care Guidelines for Diapered/Incontinent Patients served as the design frameworks.1,11 The aim of the statement of the quality improvement project was to reduce the rate of DD by 20% in the NICU.
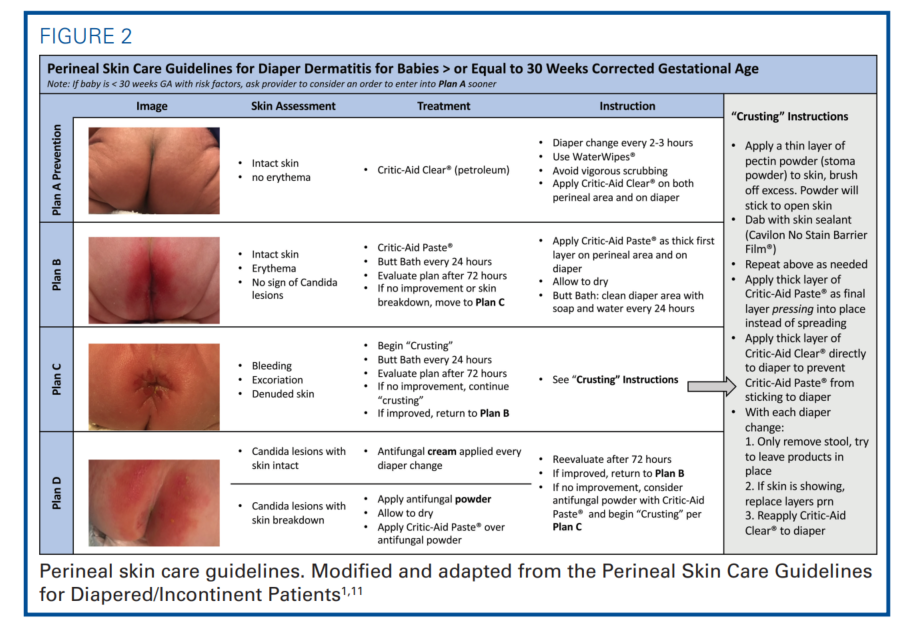
The initial steps were to standardize skin assessment and documentation. The term “rash” was a nonspecific term that described no disease severity; yet, it was commonly charted before the Perineal Skin Care Guidelines implementation. A pictorial chart was adapted from previously published guidelines to categorize DD severity.1,11 The chart assigned descriptive terminology to 4 levels of severity and recommended specific treatment plans to each (plans A-D) (Figure 2).1,11 The nurses performed a skin assessment with each diaper change and documented it with the outlined and specific terminology in the electronic medical record (EMR). To standardize a descriptive EMR reporting, nurses would select from a list of suggested terminology including rash, erythema, bleeding, excoriation, denuded, and/or others (Figure 3).
A skin assessment of nonerythematous intact skin described the absence of DD, in which the targeted plan was prevention (plan A: Prevention). Plan A stated the use of petroleum-based emollients (CriticAid Clear, [Coloplat Corporation, Minneapolis, Minnesota]) as skin-protective layers. It was the first-line barrier to all intact skin even before DD develops. Mild DD would be described as erythematous intact skin with no signs of candida lesions. The guidelines detailed the treatments for the mild DD in plan B, in which a layer with zinc oxide containing emollients (Critic-Aid Paste, [Coloplat Corporation, Minneapolis, Minnesota]) was applied to the affected area. An infant with DD would get a soap and water bath around the diaper area every 24 hours to remove irritants gently. The treatment plan C was assigned for severe DD, in which there was erythematous, denuded, excoriated, or bleeding skin without candida lesions. Plan C involved a “crusting” technique on how to apply pectin powder (Stomahesive Protective Powder, [Convatec, Oklahoma City, OK]), skin sealant (Cavilon No Stain Barrier Film, [3M, St. Paul, Minnesota]), and extra protective layers with emollients. Instruction in plan D was for fungal skin infection with candida lesions, which required antifungal treatment. When the skin lesion improved, the treatment could be de-escalated. Table 1 listed the active ingredients of the skin care emollients.
The novelty of our guideline was the use of the recently available diaper wipes, WaterWipes (WaterWipes, Portsmouth, New Hampshire). The wipes were marketed as being preservative-free, containing only 99.9% water, 0.1% grapefruit seed extract, and a trace amount of benzalkonium chloride as the surfactant agent.16 Grapefruit seed extract had been widely used as an effective antimicrobial and antibiofilm agent in the food industry.17,18 Grapefruit seed extract containing hydrogel film was shown to have antibacterial activity against Staphylococcus aureus and Escherichia coli and promote wound healing.19 However, the efficacy of grapefruit seed extract in diaper wipes on preventing DD was unknown.12
Preterm infants less than 30 weeks of corrected GA (CGA) received the same monitoring and the WaterWipes upon admission. Using barrier emollients on extreme premature infants remains controversial; emollients on incomplete epithelized skin might increase the risk for systemic absorptions and nosocomial infections.20 Therefore, preterm infants received emollients when they reached 30 weeks of CGA, except in the most severe DD cases and with physicians’ discretion.
Implementation
This quality improvement project covered 3 phases. The first phrase involved guideline design, standardizing EMR skin assessment charting, and set up data tracking. The second phase focused on staff education through multiple focus group discussions. Finally, the third phase, the implementation phase, began in January 2018.
Measures
The primary outcome measure was the incidence of DD. The infants with the same birth month were placed in a cohort and tracked over the study period. The incidences of different DD grades were compared before and after guideline implementation. Categorizing different DD grades during the preimplementation period was challenging. Before skin assessment was standardized, DD was often described as “rash” without severity grading. Mild DD (intact skin with erythema only) was not well defined prior. Diaper dermatitis, which was charted as having excoriation or bleeding under free text, was now considered as severe DD in the Perineal Care Guidelines. With uncertainty in DD grades, the incidence of severe DD during the preimplementation period might be underestimated. The duration of severe DD (skin lesions with excoriation, bleeding, or denuded) was counted as days per 100 patient-days. The patient-days were the total hospital days of all NICU infants with the same birth month.
The process measures considered the compliance of skin assessment, documentation, and the usage pattern of barrier emollients. Detailed skin assessment was necessary to follow the incidence of different DD grades and treatment responsiveness. The frequency of using the descriptive terms (erythema, bleeding, excoriation, or denuded) rather than using the nonspecific term (rash) was monitored. The percentage of infants who had emollients ordered matching the documented skin assessment was evaluated. Under skin assessment in the EMR, nurses could also document the selected skin care and treatment plan. Diaper care products ordered outside of the guidelines (eg, Domeboro [Advantine Health, Ceder Knolls, New Jersey], Desitin [Johnson & Johnson, New Brunswick, New Jersey], A&D Ointment [Bayer Corporation, Whippany, New Jersey]) were considered guideline deviations.
The guidelines introduced novel preservative-free diaper wipes. Without preservatives to prevent mold formation, the diaper wipes might pose a risk of fungal infection if used beyond the expiration date.20 The balancing measure was the rate of fungal skin infection implied from the frequency of tropical nystatin cream ordered (plan D). The cost for the different diaper wipes was also compared.
Data Analysis
Outcome measures were analyzed with statistical process control charts (QI Macros for Excel, version 2018, Denver, Colorado). The monthly incidence rates of DD grades were plotted on X-mR charts. The duration of severe DD per 100 patient-days was also plotted on X-mR charts. The control limits were set as ±3 sigma lines. Median and interquartile were used to describe GA, BW, and LOS. The MannWhitney U test and Fisher extract test were used for ordinal data or continuous data that were not normally distributed. A two-sided P value of less than .05 was considered statistically significant. Statistical analysis was performed using GraphPad Prism Version 8.3.0 for MacOs, GraphPad Software, La Jolla, California, www.graphpad.com.

RESULTS
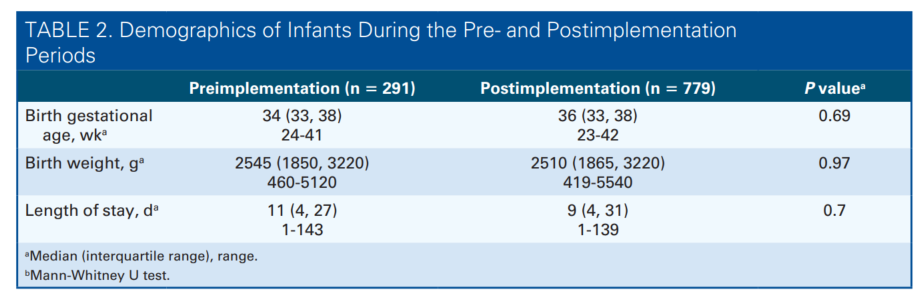
During the study period between July 2017 and March 2019, 1280 infants were admitted to the NICU and 1070 of those stayed in the NICU for longer than 1 day. There was no significant difference in GA (P = .69), BW (P = .97), and LOS (P = .70) among the infants admitted in the preimplementation (July 2017 to December 2017) and postimplementation (January 2018 to March 2019) periods (Table 2). The baseline incidence of DD was 46.0%. Infants who were born at less than 30 weeks of GA (n = 117) had a higher incidence of DD than others (67.6% vs 37.4%, Risk Ratio: 3.05, 95% CI: 2.10-4.45, P < .0001).
Outcome Measures
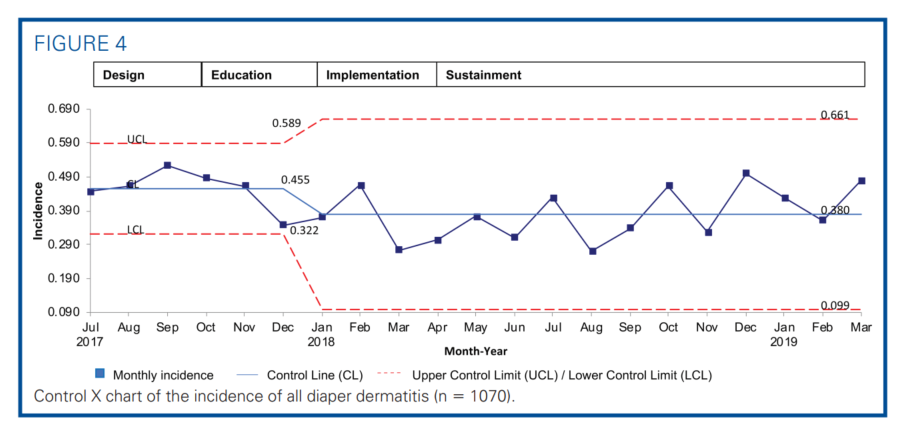
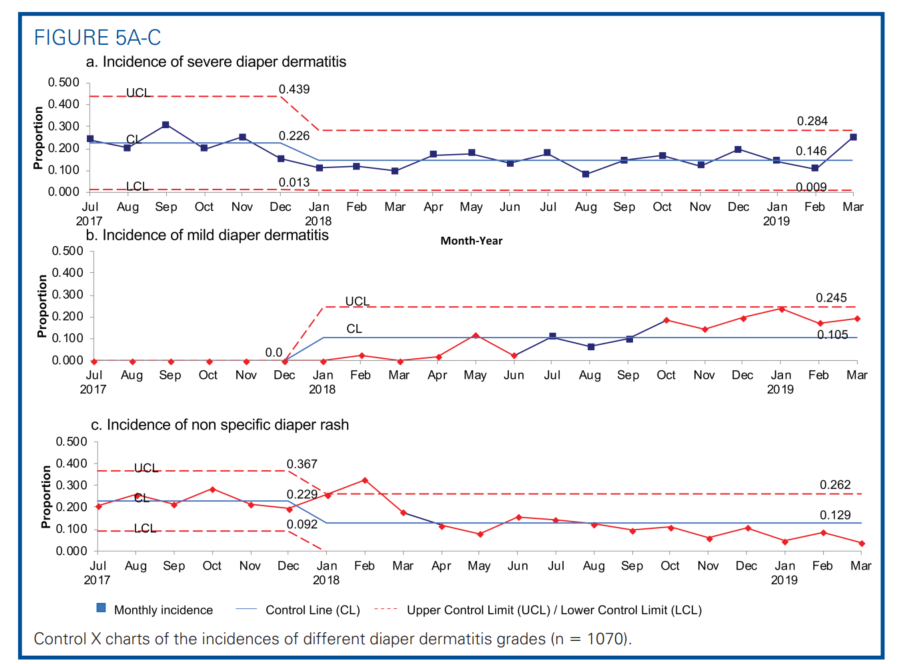
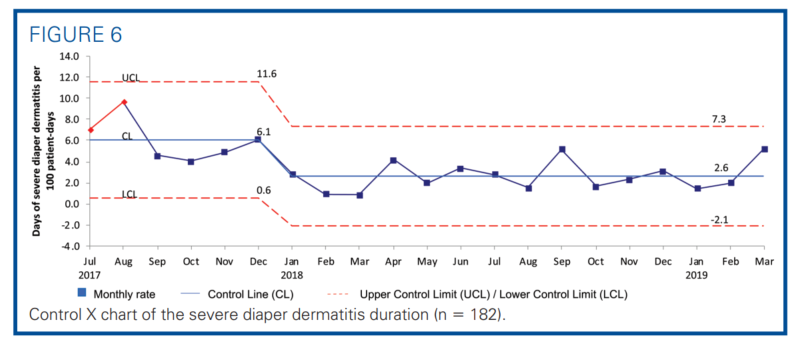
The primary outcome measure was the incidence of DD as shown in the statistical process control chart (Figure 4). After implementing the guidelines, the average incidence of DD was reduced from 45.5% to 38.0%, a 16.7% reduction. The incidence of severe DD decreased from 22.6% to 14.6%, a 34.9% reduction (Figure 5A). A rising incidence of mild DD was noted in the postimplementation period (Figure 5B). The duration of severe DD decreased from 6.1 to 2.6 days per 100 patient-days, which was a 57.4% reduction (Figure 6). All associated mR charts are shown in Supplemental Digital Content Figure 1 (control mR charts of the incidence of all DD) and Supplemental Digital Content Figure 2 (control mR chart of the severe DD duration), available at: http://links.lww.com/ANC/A63 and http:// links.lww.com/ANC/A64, respectively.
Process Measures
We also noted good process compliance. Before skin assessment was standardized, DD was often documented as “rash” without describing the severity. The use of nonspecific term “rash” decreased from 22.9% to 12.9%, a 43.7% reduction after implementation (Figure 5C). The documentation compliance correlated with increased ability to track and note more mild DD cases (Figure 5b). The preventive barrier emollients usage was not prevalent among infants before the Perineal Skin Care Guidelines implementation. Compliance with ordering preventative Critic-Aid Clear for all infants increased from 32.9 ± 16.4% to 54.1 ± 5.1%, a 64% improvement (P = .02), in the postimplementation period. Ordering Critic-Aid Paste for infants with severe DD reached 100% compliance. Ordering diaper care emollients outside of the guideline dropped from 39.0 ± 22.1% to 4.1 ± 3.0%, an 89.6% reduction (P = .01). We reported here the barrier emollients ordering pattern but not the application per each diaper change. Currently, only 9% of the diaper changes had charted the DD treatment plan chosen or applied.
Balancing Measures
The preservative-free diaper wipes might harbor fungal overgrowth if used beyond the expiration date. We observed no increase in skin or systemic fungal infections. There was instead a trend of decreased average monthly use of nystatin topical emollients (6.9 ± 2.2% vs. 3.0 ± 2.9%, P = .051) as a treatment for presumed candida fungal skin infection.
Cost Analysis
The preservative-free wipes were 6 times more expensive than the previously used brand ($0.06 vs $0.01 per sheet). Our NICU-projected annual cost increase was $7226. The novel wipes were actually cheaper than the water/cloth cleansing method. In compliance with the hospital infection prevention, only the costly surgical sponges and individual sterile water bottles could be used for the water/cloth cleansing method. The cost of the sponge and sterile water bottle would be $2.14 per day, whereas the preservative-free wipes would be $1.35 for 20 sheets of daily use.
Ethical Consideration
Institutional review board approval was obtained from The University of Utah. The committee approved a waiver of informed consent as it was a quality improvement project with minimal risk to patients.
DISCUSSION
The NICU infants are especially vulnerable to DD. Malik et al2 stated that 23% of NICU infants had appropriately documented DD. The high DD incidence (45.6%) in our NICU called for the Perineal Skin Care Guidelines development. Heimall et al1 reported a DD reduction from 24% to 11% with the implementation of evidence-based practice guidelines. We, thus, adapted the published work of Heimall et al1 while updating the available diaper care products and EMR documentation. We were able to reduce the overall DD incidence by 16.7%. The incidence of severe DD was decreased by 34.9%, along with a 3.5 days per 100 patient-days shorter duration.
Boiko9 was the first to use the acronym “ABCDE,” which summarizes the care of DD as Air, Barrier, Cleansing, Diaper, and Education. Air is for diaperfree time and air drying to reduce constant contact with irritants. Barrier is for barrier cream or emollient, which is used to provide oily protective layers from irritants and to treat already present DD. Cleansing is for washing the area with gentle rubbing and using wipes with minimal additives. Diaper is for choosing superabsorbent diapers and coupling with frequent changes. Education, finally, is for educating caretakers on diaper hygiene and treatment approaches.10 Despite the seemingly easy approaches, DD is still problematic.
Factors contributing to our accomplishment in reducing DD included early detection with improved skin assessment documentation, prevention with timely barrier emollients application, and treatment standardization. To our knowledge, we were the first to describe that the preservative-free, grapefruit seed extract wipes were well tolerated by term and preterm infants.
The key elements of our guidelines included providing pictorial descriptions for skin assessment and integrating EMR documentation, which allowed for early DD detection with the specific grading system. The incidence of DD was often underestimated due to inaccurate documentation. A study had found that over half of NICU infants were being treated with diaper care products without documented DD.10 Consistently accurate skin assessment and documentation can guide preventive strategies and gauge treatment responses.10 Skin lesions were now being described in more detail, using a precise narrative such as erythema instead of “rash.” The mild DD cases were documented more accurately and timely; hence, an increased incidence of mild DD likely reflected improved documentation. More effective treatment response tracking may prevent disease progression, as evidenced by reducing duration and incidence of severe DD.
Our guidelines standardized DD treatments. Malik et al2 reported that 5 different diaper care products were often used in the NICU, with 5.7% of infants receiving two diaper skin care products simultaneously. They also stated that no meaningful documentation was available to compare the effectiveness of those products or combinations of use; yet, stocking and ordering multiple diaper care products might accumulate a cost burden without proven efficacy.2 Our QI project streamlined the diaper care emollients. We achieved improved compliance in ordering patterns of diaper skin emollients and in treatment strategies of severe DD. We also noted improved compliance on the early application of barrier emollients to more than 50% of infants. Diaper dermatitis may further be prevented by applying barrier emollients to all infants upon admission. Our study tracked the barrier emollients ordered but not the rate of application with each diaper change. The nurses could document the treatment plan chosen (plans A-D) under “Diaper Dermatitis Protocol” with each diaper change though the increased workload or inconvenience may limit compliance.
Studies have recommended cleaning with diaper wipes, containing the least number of irritants and fragrances, as being more beneficial than using cotton wool with water.13-15 Our NICU tried out the novel preservative-free diaper wipes with grapefruit seed extract. Along with other skin care guidelines, the diaper wipes might contribute to the decreased DD. Without preservatives, the opened moist packaging would need to be replaced every 30 days due to the risk of bacterial and mold contamination.16 We found no increase in nystatin topical emollient uses or fungal skin infections. The novel diaper wipes were well tolerated by term and preterm infants. The preservative-free wipe was more expensive than other brands, but it is less costly than using sterile water and a sponge. Diaper dermatitis prevention may justify the cost of the novel diaper wipes.
Our study showed that infants born less than 30 weeks of GA had a higher incidence of DD. Our guidelines recommended applying barrier emollients only when infants reached 30 weeks of corrected GA. Further study is needed to evaluate the safety of earlier barrier emollients used on extreme preterm infants, allowing prompt skin integrity protection and treatment options.
We recognized that our study conducted in a single center lacked generalizability. But the guidelines can be easily adapted by other units. During the preimplementation period when skin assessment was not standardized, the DD grading might be uncertain and underestimating. The sustainable DD improvement over months should indicate disease modifications rather than charting changes. The cost of the diaper wipes and diaper care products might differ in other regions, affecting the cost analysis. There mightbe bias on the diaper care products’ effectiveness as the caretakers were not blinded on the products used. We also lacked information on those infants who were transferred out to the stepdown unit or other surgical units. Other confounding factors that might affect DD were not adjusted, including diet regimen, antibiotics exposure, and probiotics use.

CONCLUSION
A standardized approach to skin assessment, documentation, prevention, and treatment through the implementation of the Perineal Skin Care Guidelines reduced the incidence and severity of DD in NICU infants. Further improvement may be considered in sustaining change, empowering nurses to be proactive in ordering and escalating the treatment plan. Additional study is needed to validate whether the diaper wipes with grapefruit seed extract have proven a benefit and justify the cost burden.
Acknowledgments
The authors thank Bahr T, MD; Baserga M, MD; Gardner M, RN; Jones C, RN; Pratt C, OT, Shafter K, OT; Strevay D, RN; Tibbets V, RN; Yoder S, RN; and Warner V, RN.
References
- Heimall LM, Storey B, Stellar JJ, Davis KF. Beginning at the bottom: evidence-based care of diaper dermatitis. MCN Am J Matern Child Nurs. 2012;37(1):10-16.
- Malik A, Witsberger E, Cottrell L, Kiefer A, Yossuck P. Perianal dermatitis, its incidence, and patterns of topical therapies in a level IV neonatal intensive care unit. Am J Perinatol. 2018;35(5):486-493.
- Burdall O, Willgress L, Goad N. Neonatal skin care: developments in care to maintain neonatal barrier function and prevention of diaper dermatitis. Pediatr Dermatol. 2019;36(1):31-35.
- Pogacar MS, Maver U, Varda NM,, Micˇetic´-Turk D. Diagnosis and management of diaper dermatitis in infants with emphasis on skin microbiota in the diaper area. Int J Dermatol. 2018;57(3):265-275.
- Stamatas GN, Tierney NK. Diaper dermatitis; etiology, manifestations, preventions, and management. Pediatr Dermatol. 2014;31(1):1-7.
- Porter S, Steefel L. Diaper need: a change for better health. Pediatr Nurs. 2015;41(3):141-144.
- Esser M. Diaper dermatitis. Adv Neonatal Care. 2016;16(5):S21-S25.
- Zheng Y, Wang Q, Chen Y, et al. Shifts in the skin microbiome associated with diaper dermatitis and emollient treatment among infants and toddlers in China. Exp Dermatol. 2019;28(11):1289-1297.
- Boiko S. Treatment of diaper dermatitis. Dermatol Clin. 1999;17(1): 235-240.
- Merril L. Prevention, treatment and parent education for diaper dermatitis. Nurs Womens Health. 2015;19(4):324-336.
- Brandon D, Hil G, Heimall L, et al. Neonatal Skin Care: EvidenceBased Clinical Practice Guideline. Appendix B. 3rd ed. Association of Women’s Health, Obstetric and Neonatal Nurses. 2013;89.
- Kuller JM. Infant skin care products. Adv Neonatal Care. 2016;16(5S):S3-S12.
- Blume-Peytavi U, Lavender T, Jenerowicz D, et al. Recommendationsform a European roundtable meeting on best practice healthy infantskin care. Pediatr Dermatol. 2016;33(3):311-321.
- Visscher M, Odio M, Taylor T, et al. Skin care in the NICU patients: effects of wipes versus cloth and water on stratum corneum integrity. Neonatology. 2009;96(4):226-234.
- Vongsa R. Benefits of using an appropriately formulated wipe toclean diapered skin of preterm infants. Glob Pediatr Health.2019:6:2333794X19829186.
- WaterWipes. Ingredients. 2019 https://www.waterwipes.com/us/en/products/baby-wipes. Accessed October 13, 2019.
- Kim JH, Hong WS, Oh SW. Effect of layer-by-layer antimicrobial edible coating of alginate and chitosan with grapefruit seed extract for shelflife extension of shrimp (Litopenaeus vannamei) stored at 4°C. Int J Bio Macromol. 2018;120(pt B):1468-1473.
- Song YJ, Yu HH, Kim YJ, Lee N-K, Paik H-D. Anti-biofilm activity of grapefruit seed extract against Staphylococcus aureus and Escherichia coli. J. Microbiol Biotechnol. 2019;29(8):1177-1183.
- Jaiswal L, Shankar S, Rhim JW. Carrageenan-based functional hydrogel film reinforced with sulfur nanoparticles and grapefruit seed extract for wound healing application. Carbohydr Polym. 2019;224:115191.
- Johnson DE. Extremely preterm infant skin care: a transformation of practice aimed to prevent harm. Adv Neonatal Care. 2016:16(5S):S1-S41.
- Siegert W. Preservative trends in wet wipes. SOFW J. 2011;137:44-51.
Summary of Recommendations for Practice and Research
What we know:
- ImplementationofthePerinealSkinCareGuidelinesreducestheincidenceandseverity of diaper dermatitis.
- The approach to diaper dermatitis reduction needs to be evidence-based,
prevention-focused, and consistently practiced.
What needs to be studied:
- ExtendingthePerinealSkinCareGuidelinestotheextremepreterminfantsandproviding earlier skin integrity protection on the most vulnerable group.
- The effect of neonatal exposures to factors such as preterm milk fortifiers, liquid protein supplements, antibiotics, and probiotics on perineal skin integrity is still unknown.
What we can do today:
- Emphasizetheimportanceofprotectingskinintegrityinneonates.
- SustainthecomplianceandadherencetothePerinealSkinCareGuidelines.

Expert care for every delicate skin story
From daily diapering tips to deep dives into skin science, explore our Advice & Care guides and our dedicated Skin Care Hub!💧✨