Select your region
-
Europe
-
Americas
-
Africa and Middle East
-
Asia Pacific
A B S T R A C T
Background: Little is known on the current global prevalence of atopic dermatitis (AD) in the pediatric population.
Objective: To estimate the real-world global prevalence of AD in the pediatric population and by disease severity.
Methods: This international, cross-sectional, web-based survey of children and adolescents (6 months to <18 years old) was conducted in the following 18 countries: North America (Canada, United States), Latin America (Argentina, Brazil, Columbia, Mexico), Europe (France, Germany, Italy, Spain, United Kingdom), Middle East and Eurasia (Israel, Saudi Arabia, Turkey, United Arab Emirates, Russia), and East Asia (Japan, Taiwan). Prevalence was determined using the following 2 definitions: (1) diagnosed as having AD according to the International Study of Asthma and Allergies in Childhood (ISAAC) criteria and self- or parent-report of ever being told by a physician that they or their child child had AD (eczema); and (2) reported AD based on the ISAAC criteria only. Severity was assessed using the Patient Global Assessment (PtGA) and PatientOriented Eczema Measure (POEM).
Introduction
A previous multinational epidemiologic study in adults reported a point prevalence of diagnosed atopic dermatitis (AD) that ranged from 2.1% to 4.9% across countries.1 Although the epidemiology of AD in childhood has been the focus of several multinational studies conducted as part of the International Study of Asthma and Allergies in Childhood (ISAAC),2-6 those studies were primarily conducted between 1998 and 2004, encompassed narrow age groups (6-7 and 13-14 years), and were not necessarily representative of the country-specific populations (eg, the United States was represented by only a single study site). The prevalences reported in the ISAAC studies revealed wide variation among the centers (0.3%- 20.5%), and although the questionnaires had been previously validated, the authors suggested that the results may not be generalizable to countries in which infectious disorders with similar signs and symptoms may be prevalent.3 Wide variability in AD prevalence in pediatric populations has been reported across other studies, many of which evaluated the prevalence as part of a broader analysis, and is likely due to heterogeneity in populations, methodology, and AD clinical manifestations and nomenclature.7-14 Establishing the current prevalence and severity of AD across the pediatric age range is important for informing evidence-based medicine and for providing robust country-specific data for adequate allocation of healthcare resources for the treatment of this disease. The Epidemiology of Children with Atopic Dermatitis Reporting on their Experience (EPI-CARE) study was conducted to assess the point prevalence of AD in the pediatric population (6 months to <18 years old), including severity distribution, in countries from different geographic regions worldwide. In addition, the impact of different definitions for AD on prevalence estimates was determined. Secondary objectives of EPI-CARE, to evaluate the real-world burden among these individuals and their caregivers, will be reported in separate publications.
Methods
Study Design
EPI-CARE was a multinational, cross-sectional study designed to represent general pediatric populations across countries in North America (Canada, United States), Latin America (Argentina, Brazil, Colombia, Mexico), Europe (France, Germany, Italy, Spain, United Kingdom), the Middle East and Eurasia (Israel, Russia, Saudi Arabia [SA], Turkey, United Arab Emirates [UAE]), and East Asia (Japan and Taiwan) on sex, age, geographic region, and residential setting. The goal was to estimate overall prevalence in countries selected to cover a broad range of geographic regions while maintaining practicality of data collection that would ensure being representative of each country’s population. Data were collected according to the ethical codes of the British Healthcare Business Intelligence Association, European Society for Opinion and Marketing Research, and European Pharmaceutical Market Research Association and were compliant with the European Union General Data Protection
Regulation and the US Health Insurance Portability and Accountability Act; all subjects or their parents provided written informed consent before participation.
Collection of subject information and quality control to ensure robustness of the data were previously described in the similar multinational epidemiologic study of AD in adults.1 In short, data were collected through a web-based survey using recruitment sources that included broad-reach portals, special interest sites, and direct e-mailing.
The survey was conducted between September 26, 2018, and March 5, 2019, for all countries except Turkey and Taiwan, which were surveyed between October 7, 2019, and December 2, 2019. The questionnaire was administered in the native language of each country including validated translations of previously developed outcome measures.
Study Population
Children (6 months to <12 years old) and adolescents (12-<18 years old) in each of the countries were surveyed. Recruitment of participants was initially by parents from online panels in their respective countries. Panel members who completed the questionnaire received points redeemable for items in a prize catalogue. To reduce selection bias, panelists were blinded to the research topic when invited. Subsequent to initial recruitment, parents completed the survey for children and the parents were requested to pass control of survey completion to adolescents.
Questionnaire and Outcomes
The questionnaire consisted of 2 sections. The first section included questions that enabled the selection algorithm for families with multiple children; confirmed subject eligibility; and collected demographic information. Of note, 2 different definitions were used to estimate prevalence (Fig 1). Responders were categorized as having diagnosed AD (D-AD) if they fulfilled all items of the ISAAC criteria,2 including (1) itchy rash that was coming and going for at least 6 months, (2) had this itchy rash in the past 12 months, and (3) this itchy rash affected any of the regions including folds of the elbows, behind the knees, in front of the ankles, under the buttocks, or around the neck, ears, or eyes, and self-reported having ever been told by a physician that they had eczema. In addition, the prevalence of reported AD was estimated based exclusively on meeting the ISAAC criteria. Responders meeting the criteria for reported AD were eligible to answer the second section, which evaluated disease severity and collected information on the specialties of their physicians who are currently managing the AD, the AD treatments used, and the impact of AD on the subject and their family. Severity of AD in the past week was evaluated according to the Patient Global Assessment (PtGA),15,16 which asks, “Please check one answer that best describes the severity of your or your child’s eczema over the last week,” with responses of clear, mild, moderate, or severe. Severity was also assessed using the PatientOriented Eczema Measure (POEM),17 with a total score ranging from 0 (lower severity) to 28 (higher severity); severity groupings have been defined as bands of 0 to 7 indicating mild AD, 8 to 16 moderate AD, and greater than 16 severe AD. 18
Statistical Analysis
Quota apportionment19 was used before data collection to ensure that sampled subjects were representative of the pediatric population of the countries for sex and age (https://www.census. gov/ for all countries), geographic regions (country-specific databases), and urban vs rural except for Argentina, Colombia, Mexico, Brazil, UAE, SA, and Turkey (https://knoema.fr).19,20 If country-level quotas were not exactly met, a weighting adjustment was applied to have the structure of the total number of respondents per country match exactly the structure of the general population on the quota variables. In families with more than 1 child between 6 months and less than 18 years old, an algorithm selected the child to be surveyed based on the upcoming birthday closest to the survey, but if this child belonged to a category for which the quota had been reached, another child with the following birthday was chosen. In cases of identical birth date, the algorithm selected the child alphabetically based on the first letter of the first name.
Populations were evaluated using descriptive statistics. The 12- month prevalence was estimated for both D-AD and reported AD based on meeting the appropriate criteria.
Results
Populations
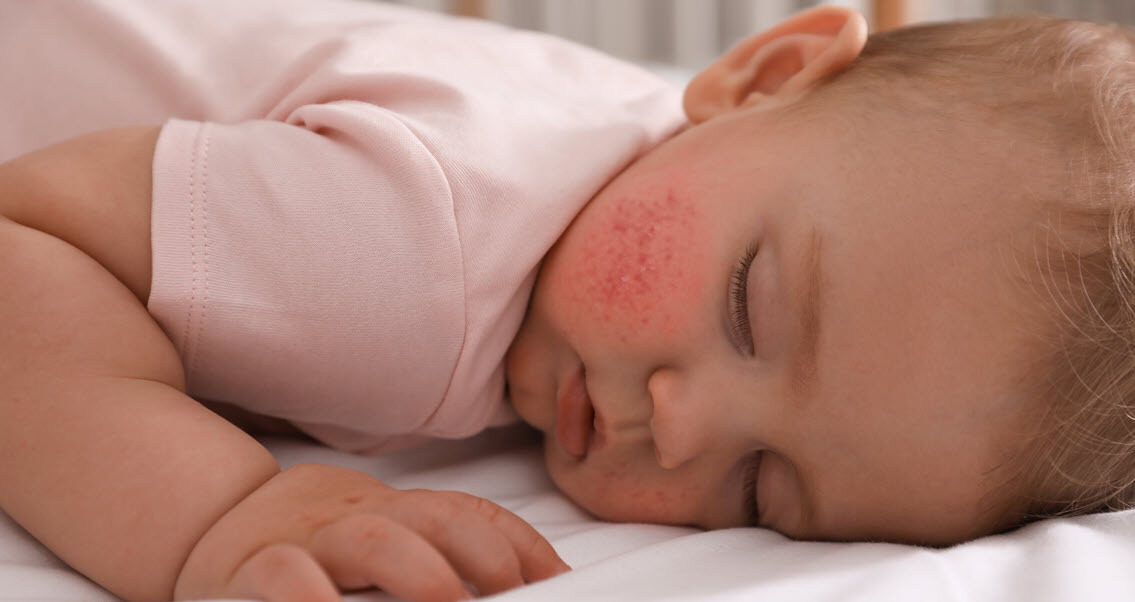
The population consisted of 65,661 pediatric subjects; 21,331 young children aged 6 months to less than 6 years, 22,238 children aged 6 to less than 12 years, and 22,092 adolescents aged 12 to less than 18 years. The demographic characteristics (Table 1) were representative of the individual countries; the regional distributions within each country were also representative (data not shown). However, residential settings seemed to be primarily driven by urban or suburban populations, although rural populations were substantial in North American and European countries (Table 1).
Prevalence
The 12-month prevalence of D-AD, based on meeting ISAAC and self-reporting having ever been told by a physician that they had eczema, revealed a substantial range across countries and within geographic regions (Fig 2A). The overall pediatric prevalence of DAD was 9.8% and 15.1% in the US and Canada, respectively, and ranged from 9.7% (Argentina) to 20.1% (Brazil) in the Latin American countries (Fig 2A). Among the countries in Europe, Germany had the lowest prevalence (8.4%), and the Southern European countries of Spain and Italy had the highest prevalence, 18.6% and 17.6%, respectively, although the prevalence in the more northerly country of the United Kingdom (UK) was also only marginally lower at 15.3%. Israel had the lowest prevalence (2.7%), not only of countries in the Middle East and Eurasia which ranged from 8.4% in Russia to 19.8% in SA but of all the countries surveyed. In East Asia, the rates were similar in Japan (10.7%) and Taiwan (11.3%).
Total D-AD prevalence was 12.1% among those aged 6 months to less than 6 years, 13.0% for those aged 6 years to less than 12 years, and 14.8% in those aged 12 years to less than 18 years. No overall pattern was observed based on age groups (Fig 2B-D), with prevalence of D-AD that ranged from 3.3% (Israel) to 18.7% (Spain) among young children aged 6 months to less than 6 years, from 2.4% (Israel) to 19.5% (Italy) among children aged 6 years to less than 12 years, and from 2.4% (Israel) to 29.4% (SA) among adolescents. Although the prevalence of D-AD was generally similar across the age groups within each country, an exception was SA, where the prevalence among those aged 6 months to less than 6 years (11.3%) was relatively lower than the 6-year to less than 12- year (18.3%) and 12-year to less than 18-year (29.4%) groups. In addition, incremental differences in prevalence from lowest to highest age groups were noted for Brazil (17.2%, 19.4%, and 23.2%), Turkey (9.6%, 12.8%, and 18.6%), and the UAE (11.7%, 15.7%, and 24.1%). Israel consistently had the lowest D-AD prevalence, which was stable across the age groups. Among the European countries, Germany had the lowest prevalence regardless of age, and among the Latin American countries, Brazil had the highest prevalence in all age groups.


When only the ISAAC criteria were considered, the prevalence of reported AD (Table 2) was higher than that of D-AD and ranged from 13.5% (Israel) to 41.9% (Italy) in the overall population; the prevalence ranged from 16.8% (Israel and the US) to 42.2% (Italy) in young children, 11.6% (Israel) to 41.9% (Italy) among those 6 to less than 12 years old, and 12.0% (Israel) to 49.6% (UAE) in adolescents. Patterns based on age and country were generally consistent with those observed for D-AD.
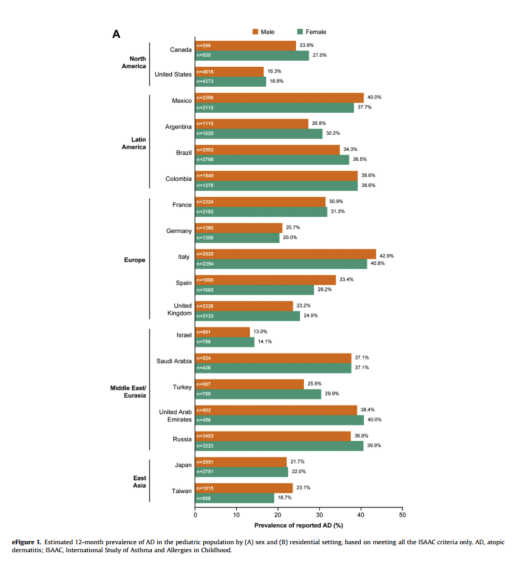
When stratified by sex, D-AD prevalence varied between males and females (Fig 3), and no clear trend was observed. Estimation of prevalence by residential setting (Fig 4) revealed that, with the exceptions of Canada and Mexico, those living in rural areas had a lower prevalence of D-AD relative to urban or suburban settings. Prevalence rates of reported AD by sex (eFig 1A) and residential setting (eFig 1B) were consistently higher than D-AD, with no trend observed for sex and lower in rural relative to urban or suburban settings except in Mexico.
Severity
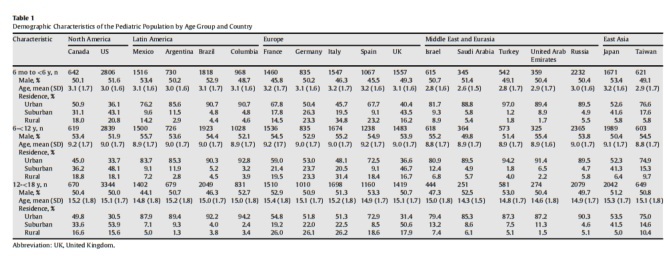
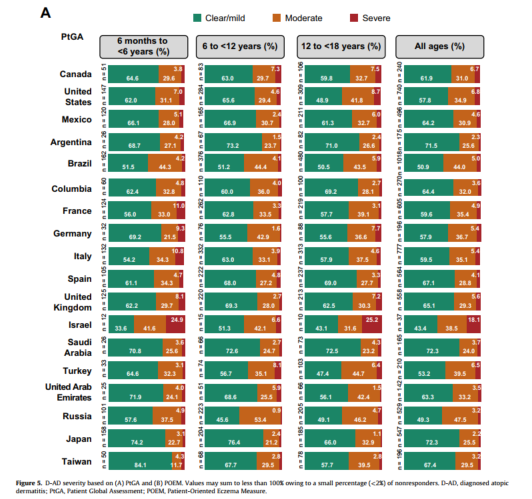
Figure 5 presents the D-AD severity distributions evaluated using PtGA (Fig 5A) and POEM (Fig 5B). Among the subjects with DAD, the proportion with severe disease was low across age groups and overall across countries regardless of which measure of severity was used. The proportion of severe D-AD ranged from 0.9% to 14.9% except in Israel, where the proportion was approximately 25% among young children regardless of the measure used and 25.2% among adolescents based on PtGA. However, the sample sizes for these age groups were small because of the low overall DAD prevalence in Israel. Considering the combined age groups, mild D-AD ranged from 43.4% (Israel) to 72.3% (SA and Japan) on the PtGA and 35.8% (Germany) to 66.1% (Spain) on POEM, with ranges of moderate AD from 24.0% (SA) to 47.5% (Russia) and 28.8% (Spain) to 55.0% (Germany) on the 2 scales, respectively (Fig 5).
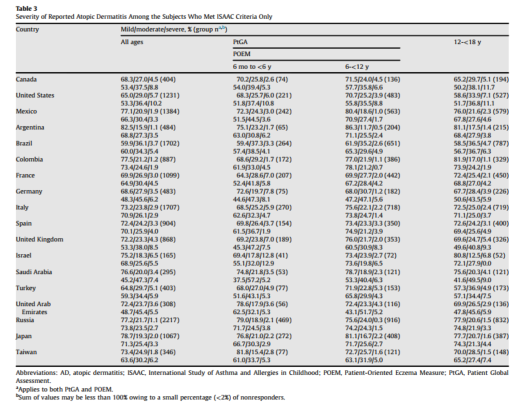
Severity distribution among those with reported AD using only the ISAAC criteria (Table 3) revealed that mild disease was generally the most frequent severity level on both PtGA and POEM. The proportion of subjects with severe disease was not only low (ranges of 1.1%-6.5% on PtGA and 1.9%-10.2% on POEM for all ages combined) but also seemed to be lower in each age group and country than among those with D-AD regardless of whether PtGA or POEM was used as the severity measure. Similarly, the proportion of subjects with moderate disease seemed to be lower than those with D-AD in the same age- and country-specific population.
Differences were observed in severity distributions between PtGA and POEM in both the D-AD and reported AD populations. These differences, which were observed across age groups and countries, revealed higher proportions with mild disease based on PtGA relative to POEM and higher proportions of moderate disease based on POEM relative to PtGA. No trends were observed regarding severity distribution across the age groups.
Discussion
The results reported here complement and expand on the previous epidemiologic study of AD in adults from several industrialized countries in North America, Europe, and Asia1 by providing the estimates of the pediatric prevalence of D-AD and reported AD in those countries and other countries encompassing different regions of the world. Sample sizes in this survey were large, consequently representing the largest multinational analysis since the ISAAC studies to evaluate AD prevalence in the pediatric population. Importantly, the accuracy of self-report was ensured by use of the validated ISAAC criteria,2 which would be expected to minimize the risk of misclassification.
In contrast to the ISAAC study,6 the present study characterized the pediatric prevalence by broad age groups including young children (6 months to <6 years old) and by disease severity, with use of demographic quotas to evaluate representative populations of the individual countries. However, the large variation between rural and urban or suburban populations suggests that, at least for some countries, the populations may have been primarily representative of the urban environment.
Results of this study reveal a wide range in the overall 12-month prevalence of D-AD in children and adolescents, with the lowest prevalence in Israel (2.7%) and the highest prevalence in Brazil (20.1%) closely followed by SA (19.8%). Such variability may be hypothesized to result from reported differences in disease onset and persistence, presentation, and diagnosis that may arise from the phenotypic heterogeneity of AD across ethnic and racial populations.21-24 It is also possible that some of this variability could be explained by cultural differences in the survey responses or discordance between symptoms and physician diagnosis that may result from disparities in access to health care.
The prevalence of D-AD, which required the combination of meeting the ISAAC criteria and having ever been told by a physician that they had eczema, was consistently lower than the prevalence of reported AD based only on the ISAAC criteria. The high prevalence rates based on patient self-report of symptoms using the ISAAC-only criteria may result from misclassification of diseases that also have symptomatic itch and involve skin flexures in children. Given the low positive predictive value of criteria that are similar to ISAAC,25 it is possible that false positives might contribute, at least in part, to the higher rates when ISAAC was used as the sole criterion; the combination of ISAAC and physician diagnosis might overcome this limitation. Simultaneously, the DAD prevalence provides an estimate of the population seeking medical advice for their condition. Thus, D-AD likely reflects disease of greater severity, as supported by higher proportions of subjects with moderate and severe AD relative to the reported AD population.


Among the countries that were previously evaluated in the adult study (US, Canada, the 5 European countries, and Japan), the 12-month pediatric D-AD prevalence across all age groups was approximately 2-fold (US) to 6-fold (UK) higher than the adult prevalence, consistent with the more frequent occurrence that has been reported in children relative to adults.26 Among the European countries, there was a generally narrow D-AD prevalence range (15.3% in the UK to 18.6% in Spain) with the exception of Germany (8.4%), and these pediatric rates are substantially higher than in the adult populations of the respective European countries.1 It is also interesting to note the difference in D-AD prevalence among the Middle Eastern countries, with SA, Turkey, and UAE not only having a substantially higher prevalence than Israel but also having prevalence rates at the higher end of the observed range across all countries. Although the Middle East has been underrepresented in epidemiologic studies of AD, the low prevalence in Israel seems to be consistent with what has previously been reported in adolescent populations from that country.27,28 Furthermore, those studies noted that Israel is characterized by waves of immigrants that reflect diverse ethnicgenetic backgrounds and that AD seems to be associated with the country of origin, suggesting genetic divergence of this disease.
Whether those observations account for, at least in part, the lower prevalence of AD warrants further study.
There did not seem to be a reduction in prevalence with increasing age as might be expected based on the often considered disease course of early onset with subsequent resolution by adulthood in most individuals.29 With few exceptions, prevalence was generally either similar across age groups or higher with increasing age, although it is possible that these observations may reflect underestimation of AD prevalence in early childhood resulting from the diagnosis not yet being established in a proportion of those in this age group. However, these observations are consistent with recent studies, including a meta-analysis of longitudinal cohort studies, which reported no clear trend in prevalence by age, with similar prevalence in childhood and adolescence.30,31 In this analysis, the prevalence of AD did not seem to have any overall trend by sex, with some countries having a higher prevalence among females, other countries with a higher prevalence among males (Argentina, Columbia, France, Italy, Spain, and Taiwan), and the US and Russia having the same prevalence in both sexes. This lack of correlation is somewhat in contrast to the ISAAC study, which suggested that among children, there was a slightly higher overall prevalence among females, although, countryspecific differences were not reported.6
The lower prevalence that was generally observed in the rural residential setting relative to urban or suburban populations provides support for a role for environmental factors in the pathogenesis of AD and is consistent with previous reports that a risk gradient among residential settings favors a lower AD risk with rural living.32-35 However, as previously noted, because urban or suburban settings were the primary drivers of the evaluated populations, further evaluation of the relationship between residential setting and AD prevalence is warranted.
As similarly observed in adult AD populations,1 mild or moderate AD was the most common severity presentations in the pediatric population regardless of age group, country, and whether assessed using PtGA or POEM. The proportions of D-AD subjects with moderate disease in the current analysis were generally higher than the 26.0% reported in a US pediatric population based on the 2007 National Survey of Children’s Health.13 These higher proportions likely reflect the criteria that were used, that is, the current analysis required meeting the ISAAC criteria and having been told by a doctor or health professional that they had eczema, whereas the other study only required the latter criterion. The presence of severe AD was consistently low, and with few exceptions, was present in less than 15% of the pediatric AD population of each country. Some disparity in severity categorization was observed between POEM and PtGA, which measure different constructs. Although POEM is based on signs and symptoms and is recommended by the Harmonising Outcome Measures for Eczema (HOME) initiative as a core measure of patient-reported symptoms,36 PtGA is a global measure that has been considered a more holistic measure of severity than clinical disease measures, because it provides a broader patient perspective and reflects lesional severity and extent, frequency of symptoms, intensity of itch and skin pain, and symptoms of anxiety and depression.15,37 When disparity between these 2 measures was present, POEM generally resulted in a slightly higher prevalence of moderate AD and a lower prevalence of mild AD than when using PtGA, as also observed in another study.38 This disparity possibly reflects POEM’s assessment of the frequency of symptoms, especially itch, that contribute to the severity rating; severity may be overestimated in mild patients with frequent symptoms on POEM relative to other severity measures. Furthermore, there may be differences in how PtGA and POEM perform with parent proxy for children less than 12 years old. In this regard, it should also be noted that there seemed to be greater consistency between the measures in the adolescent population, likely because their global perceptions match the disease activity.

A major strength of this study is its inclusion of the ISAAC criteria for identifying AD, which enables a consistent method of evaluating overall AD prevalence across countries. Another strength is addition of the criterion of “physician-confirmed diagnosis,” which helps identify the population seeking health care and enables a more robust estimate that may be potentially incorporated into models for reimbursement. Other strengths that also enhance external validity and generalizability include large sample sizes and selection of subjects that provide broad representation of the populations and regions of each country. In regard to sample size, it should be noted that this is the largest epidemiologic study subsequent to ISAAC to determine the pediatric prevalence of AD. However, limitations include a smaller selection of countries than were reported in ISAAC, especially lack of representation of African countries. Another limitation that should be considered is that some of the observed variability may have resulted from misclassification, especially because outcomes were based on self-report by subjects or their parents; such self-report, especially related to having ever been told by a physician that they had eczema, may additionally have introduced the potential for recall bias.
Furthermore, use of an online survey may represent a form of selection bias, because this method of data collection presupposes computer literacy and internet access. Selection bias may also result from potential differences between the subjects who agreed to participate and those who did not, including from subjects who opt-in to online panels and those who do not.
In conclusion, this population-based study using validated methods revealed that the 12-month prevalence of pediatric AD is higher than that reported for adult AD and displays a high variability across countries even within geographic regions. Unlike adult AD, there did not seem to be a clear association of AD with sex; prevalence among males and females varied by country. Although small differences in severity distribution were observed across age groups and countries, severe AD represented a consistently small proportion of the overall pediatric AD population (<15%). These country-level data on AD prevalence and severity may be useful in the development of public health management strategies including allocation of budgets and resources. However, these results also underscore the need for a better understanding of the factors that contribute to the observed differences across countries and regions and may provide the basis for hypothesis-driven studies to characterize these factors such as potential interactions between environment and genetics.
Acknowledgments
Medical writing support was provided by E. Jay Bienen, PhD, and was funded by Sanofi and Regeneron Pharmaceuticals Inc.
Supplementary Data
Supplementary data related to this article can be found at https://doi.org/10.1016/j.anai.2020.12.020.


References
- Barbarot S, Auziere S, Gadkari A, et al. Epidemiology of atopic dermatitis in adults: results from an international survey. Allergy. 2018;73(6):1284e1293.
- Asher MI, Keil U, Anderson HR, et al. International Study of Asthma and Allergies in Childhood (ISAAC): rationale and methods. Eur Respir J. 1995;8(3): 483e491.
- Worldwide variation in prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and atopic eczema: ISAAC. The International Study of Asthma and Allergies in Childhood (ISAAC) Steering Committee. Lancet. 1998; 351(9111):1225e1232.
- Williams H, Robertson C, Stewart A, et al. Worldwide variations in the prevalence of symptoms of atopic eczema in the International Study of Asthma and Allergies in Childhood. J Allergy Clin Immunol. 1999;103(1 Pt 1):125e138.
- Asher MI, Montefort S, Bjorksten B, et al. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC Phases One and Three repeat multicountry cross-sectional surveys. Lancet. 2006;368(9537):733e743.
- Odhiambo JA, Williams HC, Clayton TO, Robertson CF, Asher MI, ISAAC Phase Three Study Group. Global variations in prevalence of eczema symptoms in children from ISAAC Phase Three. J Allergy Clin Immunol. 2009;124(6): 1251e1258.e23.
- Jøhnke H, Vach W, Norberg LA, Bindslev-Jensen C, Høst A, Andersen KE. A comparison between criteria for diagnosing atopic eczema in infants. Br J Dermatol. 2005;153(2):352e358.
- Saeki H, Iizuka H, Mori Y, et al. Community validation of the U.K. diagnostic criteria for atopic dermatitis in Japanese elementary schoolchildren. J Dermatol Sci. 2007;47(3):227e231.
- Simpson CR, Newton J, Hippisley-Cox J, Sheikh A. Trends in the epidemiology and prescribing of medication for eczema in England. J R Soc Med. 2009;102(3): 108e117.
- Punekar YS, Sheikh A. Establishing the incidence and prevalence of cliniciandiagnosed allergic conditions in children and adolescents using routinely collected data from general practices. Clin Exp Allergy. 2009;39(8): 1209e1216.
- Belgrave DC, Granell R, Simpson A, et al. Developmental profiles of eczema, wheeze, and rhinitis: two population-based birth cohort studies. PLoS Med. 2014;11(10), e1001748.
- Ballardini N, Kull I, Soderhall C, Lilja G, Wickman M, Wahlgren CF. Eczema severity in preadolescent children and its relation to sex, filaggrin mutations, asthma, rhinitis, aggravating factors and topical treatment: a report from the AMSE birth cohort. Br J Dermatol. 2013;168(3):588e594.
- Silverberg JI, Simpson EL. Associations of childhood eczema severity: a US population-based study. Dermatitis. 2014;25(3):107e114.
- McKenzie C, Silverberg JI. The prevalence and persistence of atopic dermatitis in urban United States children. Ann Allergy Asthma Immunol. 2019;123(2):173e178.e1.
- Vakharia PP, Chopra R, Sacotte R, et al. Validation of patient-reported global severity of atopic dermatitis in adults. Allergy. 2017;73(2):451e458.
- Silverberg JI, Chiesa Fuxench ZC, Gelfand JM, et al. Content and construct validity, predictors and distribution of self-reported atopic dermatitis severity in US adults. Ann Allergy Asthma Immunol. 2018;121(6):729e734.
- Charman CR, Venn AJ, Williams HC. The patient-oriented eczema measure: development and initial validation of a new tool for measuring atopic eczema severity from the patients’ perspective. Arch Dermatol. 2004;140(12): 1513e1519.
- Charman CR, Venn AJ, Ravenscroft JC, Williams HC. Translating PatientOriented Eczema Measure (POEM) scores into clinical practice by suggesting severity strata derived using anchor-based methods. Br J Dermatol. 2013; 169(6):1326e1332.
- Deville JC. A theory of quota surveys. Surv Methodol. 1991;17:163e181.
- Flohr C, Weinmayr G, Weiland SK, et al. How well do questionnaires perform compared with physical examination in detecting flexural eczema? Findings from the International Study of Asthma and Allergies in Childhood (ISAAC) phase two. Br J Dermatol. 2009;161(4):846e853.
- Kaufman BP, Guttman-Yassky E, Alexis AF. Atopic dermatitis in diverse racial and ethnic groups-Variations in epidemiology, genetics, clinical presentation and treatment. Exp Dermatol. 2018;27(4):340e357.
- Brunner PM, Guttman-Yassky E. Racial differences in atopic dermatitis. Ann Allergy Asthma Immunol. 2019;122(5):449e455.
- Lopez Carrera YI, Al Hammadi A, Huang YH, Llamado LJ, Mahgoub E, Tallman AM. Epidemiology, diagnosis, and treatment of atopic dermatitis in the developing countries of Asia, Africa, Latin America, and the Middle East: a review. Dermatol Ther (Heidelb). 2019;9(4):685e705.
- Kim Y, Blomberg M, Rifas-Shiman SL, et al. Racial/ethnic differences in incidence and persistence of childhood atopic dermatitis. J Invest Dermatol. 2019; 139(4):827e834.
- Williams HC, Burney PG, Pembroke AC, Hay RJ. Validation of the U.K. diagnostic criteria for atopic dermatitis in a population setting. U.K. Diagnostic Criteria for Atopic Dermatitis Working Party. Br J Dermatol. 1996;135(1):12e17.
- Nutten S. Atopic dermatitis: global epidemiology and risk factors. Ann Nutr Metab. 2015;66(suppl 1):8e16.
- Wohl Y, Wainstein J, Bar-Dayan Y. Atopic dermatitis in Israeli adolescents a large retrospective cohort study. Acta Derm Venereol. 2014;94(6): 695e698.
- Shreberk-Hassidim R, Hassidim A, Gronovich Y, Dalal A, Molho-Pessach V, Zlotogorski A. Atopic dermatitis in Israeli adolescents from 1998 to 2013: trends in time and association with migraine. Pediatr Dermatol. 2017;34(3): 247e252.
- Bieber T, Bussman C. Atopic dermatitis. In: Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. China: Elsevier Saunders; 2012:203e217.
- Abuabara K, Yu AM, Okhovat JP, Allen E, Langan SM. The prevalence of atopic dermatitis beyond childhood: a systematic review and meta-analysis of longitudinal studies. Allergy. 2018;73(3):696e704.
- Abuabara K, Ye M, McCulloch CE, et al. Clinical onset of atopic eczema: results from 2 nationally representative British birth cohorts followed through midlife. J Allergy Clin Immunol. 2019;144(3):710e719.
- Schram ME, Tedja AM, Spijker R, Bos JD, Williams HC, Spuls PI. Is there a rural/urban gradient in the prevalence of eczema? A systematic review. Br J Dermatol. 2010;162(5):964e973.
- Shaw TE, Currie GP, Koudelka CW, Simpson EL. Eczema prevalence in the United States: data from the 2003 National Survey of Children’s Health. J Invest Dermatol. 2011;131(1):67e73.
- Roduit C, Frei R, Depner M, et al. Phenotypes of atopic dermatitis depending on the timing of onset and progression in childhood. JAMA Pediatr. 2017;171(7): 655e662.
- Irvine AD, Mina-Osorio P. Disease trajectories in childhood atopic dermatitis: an update and practitioner’s guide. Br J Dermatol. 2019;181(5):895e906.
- Chalmers JR, Thomas KS, Apfelbacher C, et al. Report from the fifth international consensus meeting to harmonize core outcome measures for atopic eczema/dermatitis clinical trials (HOME initiative). Br J Dermatol. 2018;178(5): e332ee341.
- Silverberg JI, Garg NK, Paller AS, Fishbein AB, Zee PC. Sleep disturbances in adults with eczema are associated with impaired overall health: a US population-based study. J Invest Dermatol. 2015;135(1):56e66.
- Silverberg JI, Gelfand JM, Margolis DJ, et al. Patient-burden and quality of life in atopic dermatitis in US adults: a population-based cross-sectional study. Ann Allergy Asthma Immunol. 2018;121(3):340e347.
A R T I C L E I N F O
Article history:
Received for publication November 3, 2020. Received in revised form December 14, 2020.
Accepted for publication December 28, 2020.
Supplementary Data


Expert care for every delicate skin story
From daily diapering tips to deep dives into skin science, explore our Advice & Care guides and our dedicated Skin Care Hub! 💧✨